Dry Eyes are a common eye condition, yet its treatment and detection has not been easy. In the past, it was treated by replacing the water component of the tear film. Pharmacies have a wall full of eye drops and all of them basically just replace the water component of the tear film.
Studies show, however, that such remedy is ineffective considering that 86% of patients with dry eyes actually have lipid deficiency caused by meibomian gland dysfunction (MGD). Thus, accurate detection and diagnosis play important roles in treating this eye condition.
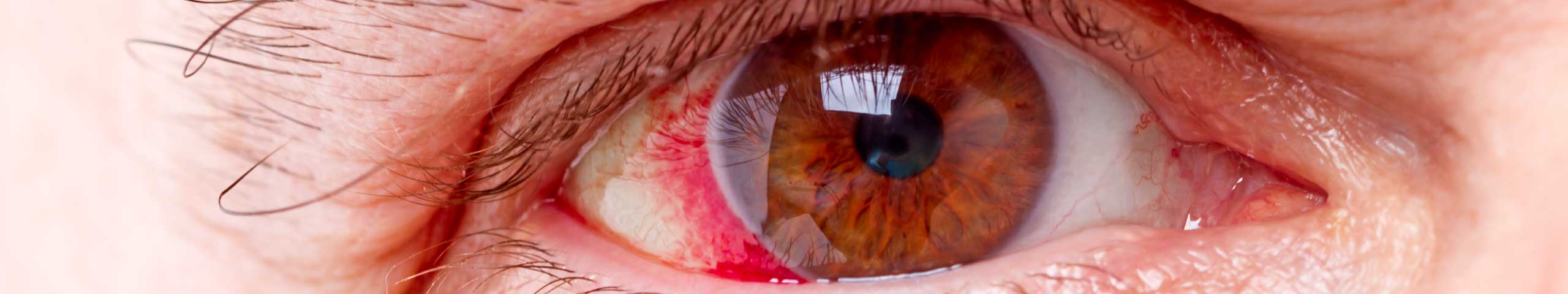
This condition is usually characterized by the following symptoms in the eyes:
- Dryness
- Burning
- Irritation
- Redness.
LipiView Interferometer
With the creation of the LipiView Ocular Surface Interferometer, detecting dry eyes no longer has to be a trial and error procedure. The LipiView is a non-invasive instrument that captures live, special and digital images of your tear film and measures its lipid content and quality. This is a standardized test that quantifies your lipid to help your eye doctor determine if you should undergo the LipiFlow procedure or other tear therapies that would treat your condition. In effect, the LipiView allows your eye doctor to visualize your tear films.
Procedure
The LipiView procedure used to detect dry eyes usually includes the following steps:
- It is done in your optometrist’s clinic.
- It only takes about five minutes.
- Your doctor will ask you to look into a camera and blink normally.
- The LipiView Ocular Surface Interferometer, a special light camera, and a computer program will take care of everything else.
Other Procedures
Apart from the LipiView procedure which assesses the quality of your tear film and your blink dynamics, your eye doctor may also opt to conduct a meibomian gland evaluation. These procedures will help your doctor assess your blink and tear mechanics which are necessary to determine the appropriate treatment for your dry eye condition.
These procedures may be done to diagnose a patient’s condition prior to an eye surgery and to manage patients who develop this condition after an eye surgery.
Reliability and Safety
The LipiView Ocular Surface Interferometer was created by TearScience, Inc., which is a privately-held medical company. This North Carolina-based company pioneered devices that provide vast clinical improvement in the treatment of evaporative dry eye condition. The technology is safe and reliable so you have nothing to worry about.
The LipiView Technology was even given the Medical Design Excellence Award by UBM Canon in 2011. UBM Canon is a leading business-to-business media company that is dedicated to the advanced manufacturing sector. This competition is the only awards program that exclusively recognizes advances and contributions in the design of medical products that are cleared for marketing by the United States Food and Drug Administration. Entries to this competition are evaluated based on design and engineering features.
The LipiView Ocular Surface Interferometer is highly-reliable as it provides a quantifiable alternative for evaluating a patient’s tear film. It does away with the highly-subjective tests that eye doctors used to rely on to evaluate patients with dry eye conditions.
The first LipiView Interferometer in Australia is being installed at The Eye Practice (Sydney CBD). Any questions can be directed to The Eye Practice on (02) 9290 1899 or click the QUICK ENQUIRY Tab at the top right hand side of this website.